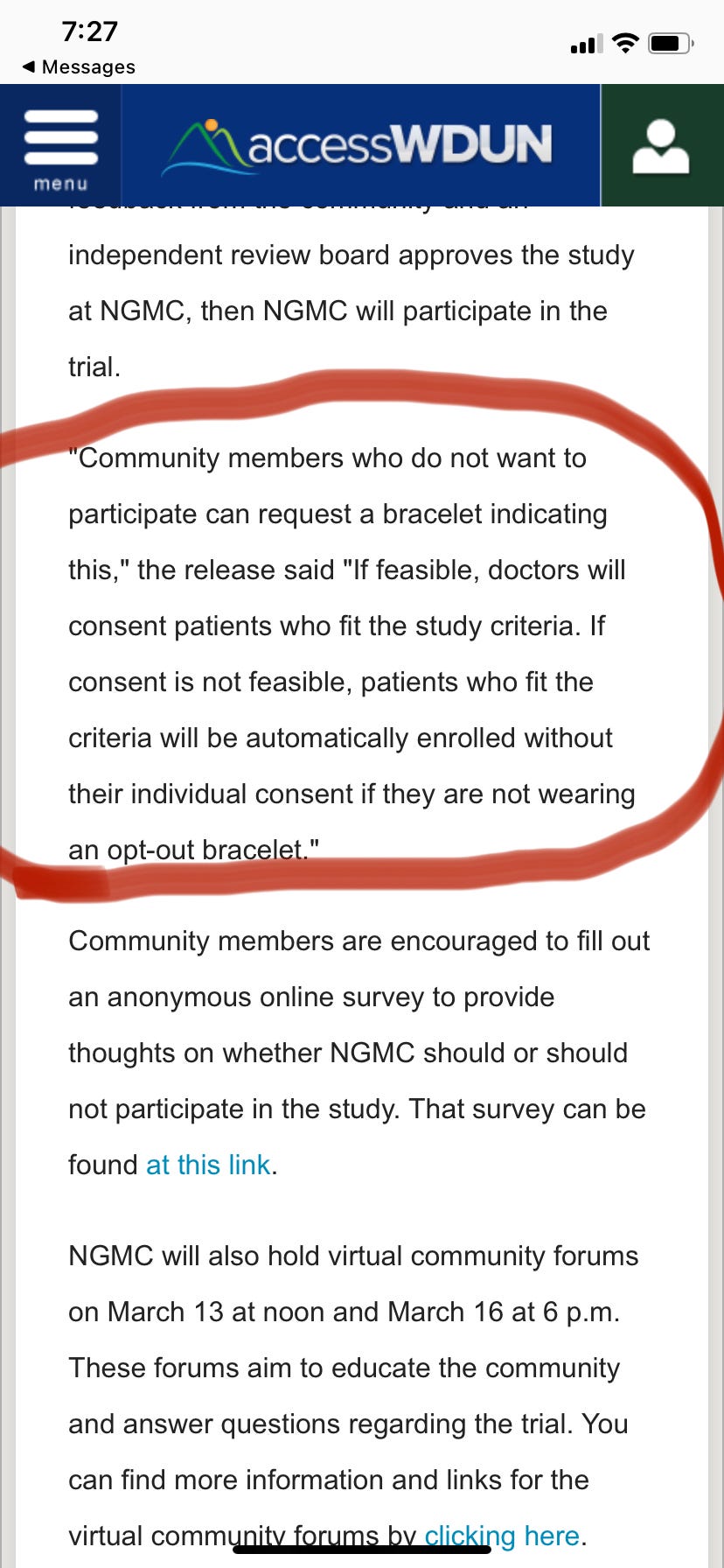
When was the last time your community automatically enrolled you in a clinical trial, requiring you to wear a bracelet to “opt out?” That’s exactly what is going on with NCT05568888. A nurse in our local advocacy group brought this to my attention. It didn’t sit right with her. What about informed consent?
On the surface, it all sounds great. It is to save lives. I have no doubt that the clinical rationale is appropriate and designed to save lives. But, isn’t it amazing that we’ve gone this long without ever having to wear a bracelet to indicate our medical preferences and “opt out” of a medical experiment?
This attack is subtle—a nudge. I’ve reached out to medical freedom folks and people in healthcare to ask, “do you see something wrong here?” I’ve received responses like, “it’s an FDA approved drug. If peoples’ lives can be saved, they should have access to the drug.” To which I say, “I agree.” In the past, I would have probably just chalked it up to, “there are regulatory systems in place, I’m sure everything is on the up and up.” The alarm on my B.S. meter is now dialed way down to 0.5 on a 5 point scale, I don’t buy it.
So, I fight off the voice that says, “you don’t know—you aren’t qualified.” First of all, it is usually the person that seems the “least qualified” that actually asks the best questions—so we all need to be courageously fighting off that voice and ask the darned question. Then, I ask myself truly, what do I know of the subject? Well, I took a research methodology class in graduate school in 1995. I am familiar with terms like prospective and retrospective studies. Then I dig out my training records from my stint working in clinical trials. On December 8, 2017, I “completed the NIH Web-based training course ‘Protecting Human Research Participants’” states the certificate issued by The National Institutes of Health (NIH) Office of Extramural Research. Oh, there are a bunch more, including “Informed Consent in Clinical Trials of Drugs, Biologics, and Devices,” completed on January 5th, 2018. Now I become emboldened. I may not know much, but I know enough to be dangerous…to the enemy!
No one yet has been able to answer me the question of how in the past emergency room procedures were changed without this research methodology? I suspect that at the heart of it all is the fact that an approved FDA drug has always been allowed to be used “off label” if the licensed provider has a good rationale that the therapeutic could be an effective treatment for a different condition. We certainly know that there has been an attack on this core medical principal as of late. There are witch trials being conducted in state medical boards across the country as we speak. Let’s count that as one motivation for this nudge. We must have people believing that all approved drugs are required to go through prospective randomized trials before they can be “approved for use” in treating other conditions. Otherwise, we may not be able to guarantee that the next power grabbing, money making opportunity that presents itself will be “solved” by our pharma gods creating new therapeutics. That IS the world order!
Second, the evil in the world really really likes the idea of health passports. How else will we know that someone is “safe” to be around if they aren’t displaying their health status using something like a bracelet? Yup, I call this a NUDGE.
Third, informed consent hasn’t really existed for decades. This little ploy in research methodology just normalizes it so we won’t even question when the next medical experiment is thrust upon us. They have already neutered the Nuremburg Code.
Alright smarty pants, so what DO you propose? Do what I believe they have always done. Give the approved drug to those that meet the clinical criteria, monitor outcomes and report it as a retrospective study. Alter clinical protocols as data determines is appropriate.
So, what are WE to do? First, check the alarm settings on your B.S. meter. For too long, we’ve been content to have it rest between 3.5 and 4 on a 5 point scale. Now that the B.S. is piling up to a level 4+, more of us are aware that lots of things smell really crappy. Have courage—ask the questions and question everything. Ask Holy Spirit for discernment when it comes to making decisions for yourself and your family. Deception is truly running rampant, just as the Bible warned us it would.
Turning to the Bible for guidance on courage, I plugged the keyword into one of my favorite sites: https://www.gotquestions.org/. Not sure how I missed that courage is the opposite of fear? I guess because I don’t think that the absence of fear always requires courage. Regardless, the site directs us to many passages for Bible study.
Let’s close with this promise from Proverbs 3:25-26 (NLT) You need not be afraid of disaster or the destruction that comes upon the wicked, for the Lord is your security. He will keep your foot from being caught in the trap. Jesus tells us in Matthew 12:30 (NLT), “Anyone who isn’t helping me opposes me, and anyone who isn’t working with me is actually working against me.” So, we don’t have to fear the wicked. But apathy, going along to get along, isn’t going to cut it. We all have to make a choice. I pray that you will hold your sword & shield, stand courageously for your family & community, and encourage fellow soldiers in this battle against evil!
Post Script exercise:
I did have to reach out to the study coordinators of the above study to ask for the correct NCT# (NCT05568888). It should be standard practice for that to be reported with any public outreach to enroll clinical trial participants—but it isn’t.
As I did my initial search trying to find the study in clinicaltrials.gov, this other clinical trial regarding Severe Traumatic Hemorrhage popped up, NCT05738642. It isn’t yet recruiting and was initially posted February 23, 2023. Interesting that the sponsor is listed as, “Affiliated Hospital, School of Medicine, Zhejiang University.” I’m curious if this too will be allowed to waive informed consent? Reading the “Exclusion Criteria,” I’m not sure how they could rule out the conditions without talking to the patient or a family member first? Isn’t that the argument in the above study for waiving “informed consent” and making you wear an opt out bracelet—no time in life or death situations?
“ (1)history of anticoagulation and antiplatelet drugs (2) has a deep venous thromboembolism (VTE) and pulmonary embolism (PE) history (3) (4) on patients with severe head injury occurred cardiac arrest patients (5) lactation, pregnant women 6 Abdominal cavity viscera perforation surgical treatment in patients with 7) in patients with sepsis shock patients with all kinds of mental illness incapacitated pet-name ruby are unknown agent for informed consent.”
(Don’t ask me what “mental illness incapacitated pet-name ruby” means, an internet search of the complete phrase didn’t turn up anything.)
Alarm bells going off regarding “biomedical research” yet? Can we really afford to stand down and let ourselves be nudged?